
In some cases, the resulting mineral formula is modified by assigning the cations to their probable crystallographic sites based on their probable coordination numbers.īecause electron beam analyses do not differentiate among the valence states of iron (iron is normally reported as FeO), the formula calculation procedure may be different for different iron-bearing minerals. Multiply the "normalized oxygen numbers" of each oxide by the number of cations per oxygen in the oxide formula.Multiply the resulting "oxygen number" of each oxide by a normalization constant (equal to the number of oxygens in the desired formula divided by the sum of the "oxygen numbers").Multiply the resulting "mole number" of each oxide by the number of oxygens in the oxide formula.Divide the weight percentage of each oxide by the formula weight of that oxide.The basic steps for formula calculation are: Retrieval of standard chemical formulas for minerals from chemical analyses is an exercise in (a) conversion of units of quantity and (b) normalization of sums to match the commonly used formula conventions. This page provides information and tools for the calculation of minerals formulas from chemical analyses. They obscure relationships that may be obvious when the compositions of minerals are expressed in terms of formulas (atomic proportions). Weight percentages are not the most convenient format for many uses of mineral analyses. 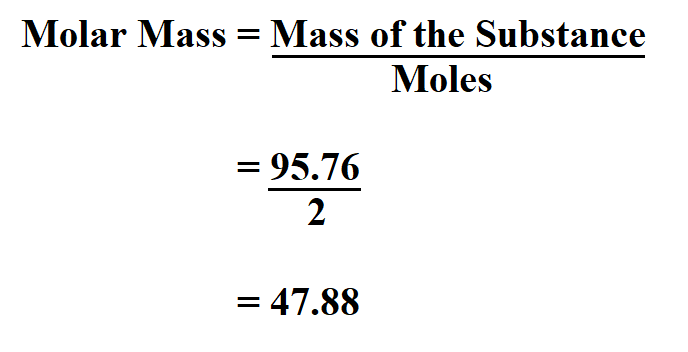
It is assumed that each mineral is electrically neutral and that the positive charges on the cations are balanced by an appropriate quantity of oxygen anions. Although little weighing is involved in most modern chemical analyses ( electron microprobe or energy dispersive x-ray spectrometer) weight percentages are reported for historic reasons (chemists performed their analyses using gravimetric techniques). Note: You can use other related calculator Chemical Equation Balancer.John Brady, Smith College and Dexter Perkins, University of North Dakota What is it?Ĭhemical analyses for silicate minerals are commonly reported in weight percentages of the oxides of the elements determined. 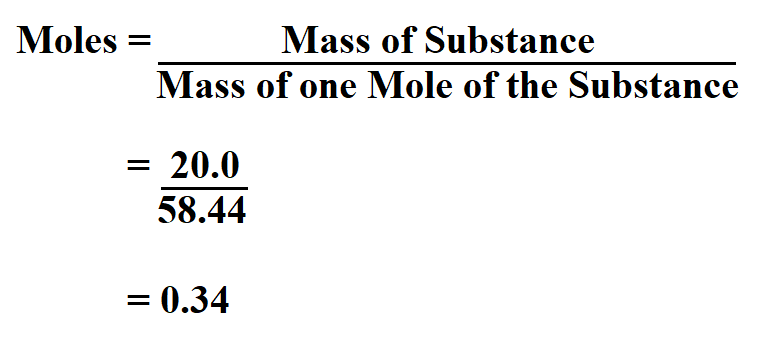
Therefore the polyatomic ion for the above positive and negative ions is Lithium Chloride. The hydroxide cation (OH-) and the phosphate cation (PO3− 4) are both polyatomic ions.īarium has a +2 charge and hydroxide has a -1 charge, thereforeġ Ba 2+ ion is required to balance 2 OH- ions Ammonium has a +1 charge and phosphate has a -3 charge, thereforeģ NH 4 + ions are required to balance 1 PO 4 3- ion Potassium has a +1 charge and sulfate has a -2 charge, thereforeĢ K + ions are required to balance 1 SO 4 2- ionĬalculate the polyatomic ion for the given positive and negative ions. Polyatomic ions consist of multiple atoms that combine with other ions to form an ionic compound. In an ionic compound, electrons are transferred to atoms and molecules imparting a charge on both the donor and recipient of the electron.Ī polyatomic ion considers two or more atoms at a time as constituents. Phosphate - PO 43- Cation and Anion calculatorĪ polyatomic ion, also known as a molecular ion, is a charged species (ion) composed of two or more atoms covalently bonded or of a metal complex that can be considered as acting as a single unit in the context of acid and base chemistry or in the formation of salts.īy putting the positive ion and the negative ion in the respective boxes, you can find the scientific name of that polyatomic by using ion calculator. 
Write down the information you have for the charges of the component ions and balance them to answer the problem.īicarbonate (or hydrogen carbonate) - HCO 3. When there are two or more polyatomic ions in a formula, enclose the polyatomic ion in parentheses. When you write the formula for an ionic compound, remember that the positive ion is always listed first.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
